The ISMRM Impact Project
The True Impact of Research Published in the ISMRM’s Journals
BY: TOM GRIESLER, UTSAV SHRESTHA, JULIUS HEIDENREICH, FRANCESCO GIGANTI, DAIKI TAMADA, ANDRADA IANUS, DIEGO HERNANDO
Most people will never read a scientific paper or attend a medical research conference. Yet many of us benefit from biomedical research without ever realizing it. If you’ve had an MRI exam — to diagnose a sports injury, examine your brain, or monitor a disease — you’ve relied on decades of research that quietly shaped how that exam was performed, how long it took, and how clear the images were.
For researchers, it is not always clear when that impact happens. New ideas are tested, refined, and eventually published, often years before they reach hospitals and clinics. Traditionally, scientists measure the influence of this work using academic metrics: how often a paper is cited by other researchers, or how prominent the journal is. These metrics help track scientific discussion, but they say little about how research ultimately affects patients’ lives.
This gap is especially visible in medical imaging. The global MRI research community is relatively small; its main scientific gathering — the ISMRM Annual Meeting & Exhibition — brings together around 5,000 specialists each year. These are the people most likely to read and cite papers published in the Society’s journals, Magnetic Resonance in Medicine (MRM) and Journal of Magnetic Resonance Imaging (JMRI), resulting in modest citation numbers compared to other scientific fields.
And yet, MRI itself has become a routine part of modern medicine. Ask friends or family whether they’ve had an MRI scan in the past decade, and many will say yes. Importantly, these exams are often essential for guiding care — helping diagnose disease, plan treatment, or monitor recovery. Many of today’s faster, safer, more informative MRI exams would not be possible without research first shared at ISMRM conferences and published in ISMRM journals.
In this occasional series, we look back at MRI innovations that had a major impact on clinical practice. To illustrate this impact, we trace how ideas moved from early conference abstracts to scientific papers, to widespread use in hospitals, and how they improved real-world patient care.
Our first article highlights one of the true cornerstones of modern, fast MRI: parallel imaging. Few developments have had such a broad and sustained impact on clinical practice. Remarkably, this entire evolution — inception, breakthrough discoveries, and clinical dissemination — has unfolded within the ISMRM community, from the first conference abstracts to pivotal papers and rigorous clinical evaluation. Christiane Kuhl, MD; Tom Grist, MD; and Howard Rowley, MD were interviewed and describe first-hand experiences of how parallel imaging was adopted and integrated into routine clinical MR imaging.
From Abstract to Paper to Product
Whether you’re an MRI expert or not, it’s unlikely that you’ll need to be convinced that MRI is a fascinating technology. It can visualize nearly any anatomy with unparalleled soft tissue contrast — and it accomplishes this without the need for harmful radiation. However, one persistent challenge has troubled researchers and clinicians: MRI is slow. Instead of capturing the whole picture at once, an MRI system typically performs many separate measurements, one after another, until there is enough information to reconstruct an image. This results in an inherent tradeoff between spatial resolution, signal-to-noise ratio (SNR), and acquisition time — sometimes called the “vicious circle of image quality” or “MRI triangle”. In essence, this means that you can’t achieve high-resolution, high-SNR images quickly; at least one factor must be compromised. While this tradeoff still exists, decades of innovation have steadily shifted the triangle in a more favorable direction. Today’s MRI systems can achieve combinations of speed, resolution, and image quality that were once thought impossible.
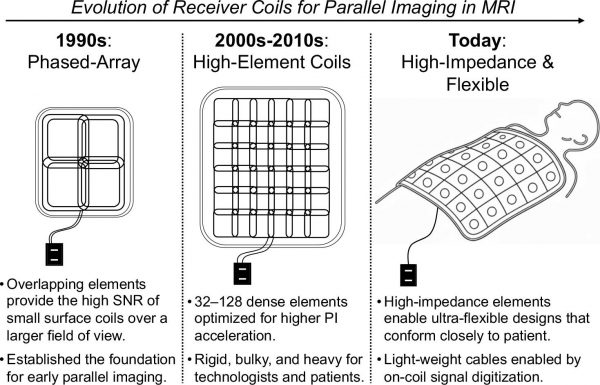
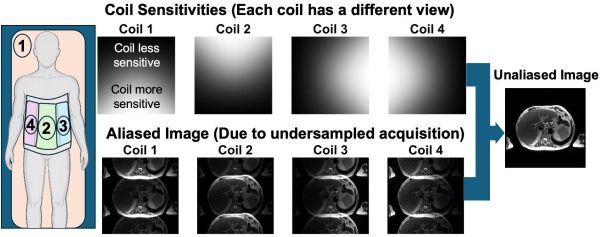
An early milestone in this technological shift was the “NMR phased array” introduced by Pete Roemer and colleagues (MRM, 1990). Roemer demonstrated that you could use multiple receiver antennae (“coils”) at once — designed so they don’t interfere with each other — and combine their signals to get substantially higher SNR over a large field of view without longer acquisition times or lower spatial resolution. While Roemer’s work was mainly focused on image quality and coverage, rather than speed, it established a crucial ingredient: each coil “sees” the body a little differently. By the late 1990s, several groups independently realized that these coil-to-coil differences could be used not just to improve SNR, but also to reconstruct images from less acquired data — shortening acquisition times. Parallel imaging was born.
One of the first concrete implementations of parallel imaging acceleration arrived in 1997 with the introduction of SMASH — short for Simultaneous Acquisition of Spatial Harmonics. Authored by Dan Sodickson and published in MRM, the paper demonstrated for the first time how the coils’ different spatial “views” could be used to mathematically replace some of the measurements MRI would otherwise have to acquire one-by-one. In this way, SMASH enabled faster MRI without a proportional loss in image quality. SMASH was also shared with the community at that year’s ISMRM meeting in Vancouver. However, as is sometimes the case with major innovations, its significance was not immediately recognized. Although SMASH received considerable scientific attention, persuading industry partners of its broader impact took additional effort, as Sodickson later recounted in a 2018 interview with MRM Highlights. Only when competing parallel imaging approaches began to emerge did momentum in the field truly start to build.
Among those inspired by Sodickson’s 1997 ISMRM presentation were Klaas Pruessmann and Markus Weiger. The story goes that, while decompressing after the meeting on a canoe trip, they talked through ideas and sketched a different path that they would later call SENSE (Sensitivity Encoding), presented at ISMRM just one year later and published in MRM in 1999. At a high level, the contrast with SMASH was this: SMASH aimed to use the coils’ different “views” to recreate some of the missing measurements MRI would normally collect, whereas SENSE moved the problem to the image itself — accepting that faster acquisition causes signals from different parts of the body to overlap in the image, which could be mathematically “unmixed” using the additional coil information. That shift offered a direct and broadly applicable framework for reconstruction. In Sodickson’s recollection (as discussed in the aforementioned MRM Highlights interview), the competition between SMASH and SENSE was ultimately beneficial: it sharpened the field’s focus, generated wider interest, and helped drive both academic attention and eventual industry adoption and clinical dissemination.
Another major milestone followed a few years later with GRAPPA (Generalized Autocalibrating Partially Parallel Acquisitions), introduced by Mark Griswold and colleagues in MRM in 2002. Similar to SMASH, from which it evolved, in GRAPPA the MRI system deliberately skips some of the usual measurements to save time. Now, however, it also acquires a small amount of built-in reference data during the same acquisition. From that reference data, GRAPPA “learns” how the different coils’ signals relate to each other, and then uses that learned relationship to synthesize the missing measurements before forming the final image. Compared with SENSE’s strategy of unmixing overlap in the image, GRAPPA’s appeal was that it could often achieve similar acceleration without requiring additional acquisitions to obtain the necessary coil information. This helped make parallel imaging easier to deploy across a wide range of clinical protocols.
Within just a few years, parallel imaging had gained major scientific momentum, and captured the attention of the MRI industry. While translating academic ideas into clinical products is often a slow process, commercialization in this case moved unusually quickly. The commercial introduction of parallel imaging became a textbook example of the value of close collaboration between university researchers and industry partners. At the time Klaas Pruessmann and his group at ETH Zurich published the SENSE method, they were already working with Philips, enabling them to become the first manufacturer to make parallel imaging widely available on clinical MRI systems around 2000. Other major MRI vendors quickly realized they needed to follow to remain competitive. Siemens Healthineers introduced its iPAT technology the same year, initially offering a mix of SENSE-like and autocalibrating methods and later placing strong emphasis on GRAPPA through collaborations that included Mark Griswold. GE Healthcare introduced ASSET, a coil-sensitivity–based implementation, and later added an autocalibrating extension of GRAPPA known as ARC. By 2002, Toshiba Medical Systems (now Canon Medical Systems) had entered the field with its own implementation, SPEEDER — marking parallel imaging’s broad adoption across the major MRI vendors. Toshiba/Canon would later include a GRAPPA-like autocalibrating implementation known as Exsper. Finally, by the time United Imaging entered the MRI market in the 2010s, parallel imaging had become ubiquitous and was therefore integrated by default into its workflows.
Real World Impact: Clinical Dissemination
In clinical practice, parallel imaging is chiefly employed to address three key challenges: prolonged acquisition times, patient motion, and image artifacts. While often mainly described as an acceleration method, parallel imaging did more than make MRI faster: it changed what MRI could realistically do in clinical practice, and who could benefit from it. Clinicians recognized its value early because the benefits were obvious and directly relevant. Notably, parallel imaging is not tied to a specific acquisition type or body part – instead, it can be integrated with various techniques and speed up a broad range of MRI examinations.
To illustrate how parallel imaging made its way into clinical practice, we interviewed three internationally recognized academic radiologists and clinical leaders who witnessed and shaped its adoption from different clinical perspectives: Christiane Kuhl (breast imaging), Howard Rowley (neuroimaging), and Tom Grist (cardiovascular imaging). Their reflections provide first-hand perspectives on how parallel imaging reshaped clinical workflows, technical expectations and standards of image quality.
Howard Rowley recalled that seeing diffusion-weighted imaging performed with parallel imaging acceleration was an immediate relief. The technique dramatically reduced geometric distortions and improved image quality. Importantly, parallel imaging did not impose a single trade-off. Instead, it gave clinicians flexibility: the gained acceleration could be used for shorter acquisition times, higher spatial resolution, or reduced artifacts. Rowley likened this flexibility to “money in the bank”, ready to be invested where it mattered most.
Beyond improving image quality and robustness, parallel imaging also expanded access to MRI. By shortening acquisition times and reducing sensitivity to motion, it made MRI feasible for patients who previously struggled to tolerate long or demanding examinations. This includes children, elderly patients, and patients with pain, neurological disease, or limited ability to remain still or hold their breath.
In some body regions, parallel imaging was not only an incremental improvement, but a prerequisite for clinical viability. Breast MRI is a prominent example, as Christiane Kuhl emphasized – she described the introduction of parallel imaging as “a game changer for breast imaging”. By resolving the long-standing trade-off between spatial and temporal resolution, parallel imaging helped establish breast MRI not only as the most sensitive diagnostic modality for breast cancer detection, but also as a viable screening tool in high-risk populations.
Similar gains played out across many other applications. In cardiac MRI, parallel imaging allowed rapid image sequences of the beating heart to be acquired in a shorter breath-hold, enabling reliable evaluation of heart motion even in patients with limited breath-holding capacity. In cancer imaging, especially during contrast-enhanced exams, faster acquisitions made it easier to reliably capture the brief early moment when a tumor first becomes visible as the contrast agent arrives during arterial phase enhancement. Likewise, parallel imaging improved the ability of dynamic MR angiography to capture the arterial phase of enhancement and allowed larger volumetric spatial coverage during a limited breath-hold time. In musculoskeletal MRI, time savings were often reinvested into higher spatial resolution, enabling more detailed visualization of small anatomical structures. Today, parallel imaging acceleration is so deeply embedded in routine MRI protocols that it often goes unnoticed – yet it fundamentally changed expectations of what clinical MRI can deliver.
Importantly, the close collaboration between clinicians and those developing MRI technology was essential to the rapid clinical dissemination of parallel imaging. As noted by Tom Grist, the deployment of parallel imaging acceleration benefited from the presence of many physician-scientists who were directly involved in understanding new MRI methods and helping to bring them into clinical use. Although the mathematical foundations of parallel imaging were complex, its clinical benefits were easy to appreciate, which helped build trust and speed up adoption.
In this context, Christiane Kuhl and Tom Grist both emphasized that the close interaction between clinicians and researchers/physicists is what made — and continues to make — ISMRM a uniquely collaborative scientific society. Dr Kuhl notes that while researchers must learn about real-world clinical challenges from practitioners, clinicians also need a solid technical understanding of the imaging modality they use. She compares this relationship to a pianist and an instrument: even the finest instrument cannot produce great music if the musician does not understand how to use it.
Current and Future Developments
Meanwhile, MRI research within the ISMRM community keeps pushing the MRI triangle for the benefit of patients worldwide. Since the introduction of parallel imaging acceleration, newer methods have been developed to enable further acceleration. Simultaneous multi-slice imaging allows the acquisition of several imaging slices at once, increasing coverage without extending acquisition times. Higher dimensional MR applications (for example, 4D MRI, which captures three spatial dimensions over time) have benefited from increased data redundancy to enable further acceleration. More recently, artificial intelligence-based methods have enabled even faster imaging while maintaining high resolution and image quality.
Following in the footsteps of parallel imaging pioneers, the ISMRM community keeps driving innovation. By working across disciplines including clinicians and scientists, we have an opportunity to continue the ISMRM’s tradition of clinical impact while tackling some of the world’s most pressing healthcare challenges.