By Derek Jones
Professor Joanne Ingwall pioneered the use of phosphorous magnetic resonance spectroscopy to study cardiac energetics. She played a major role in shaping the ISMRM, introducing initiatives and frameworks that persist to this day, and is a keen advocate of mentoring students, fellows and junior faculty. On Friday 5th April, during the peak of the ‘SuperBloom’ in her home town of Borrego-Springs, Southern California, Professor Ingwall kindly agreed to chat, over the internet, to Derek Jones about her life in NMR and her role in the society, including the recognition of spectroscopists and her love of conference ribbons.
DKJ: Professor Joanne Ingwall, welcome to MRM Highlights.
JI: Thank you. I’m delighted to be here.
THE FORMATION AND EARLY DAYS OF THE SMRM AND ISMRM
DKJ: Many people will be reading this interview at the ISMRM in Montreal, and a good proportion of those will be attending the meeting for their very first time. Before we explore your story, could you cast your mind back to your very first meeting, of what was then called SMRM? What are your recollections of those early days?
JI: Well, I probably attended the first SMRM meeting in Boston in 1982. The group of people that formed the society was based there. Gerry Pohost loves to organize things like this, and spent a lot of energy pushing for it, together with Tom Budinger, Alex Margulis, Paul Lauterbur and Britton Chance. They also invited me and Kate Scott, who was in Florida at the time. Tom Budinger decided that we had to be identifiable as members of the board and had to wear big name badges – horrible things with ribbons coming out. Oh, it was awful. I nearly died.
DKJ: The tradition of board ribbons has continued to the present day!
JI: Really? I think that’s awful. I understand the rationale, but it’s a bit much, it’s like a horse!
DKJ: “Best in Show”?
JI: Exactly! But I also remember being amazed that there were so many people who came out of the woodwork, even though the field was exceedingly new, and how many people actually showed up year after year.
I remember the New York meeting best of all, I suppose because it was fun to go to New York. I’ll tell you a funny aside. The meeting was held at the New York Hilton; we were their largest client by the time the 89 and 90 meetings came around. When I was president, they sent a car to the airport to get me and my husband. We get in this limousine and we’re driving through Central Park, and my husband turns to me and said, “I don’t understand this. I thought limos were only for important people.” I nearly killed him!
DKJ: So this was SMRM, not ISMRM. Many people reading this interview won’t be familiar with the backstory of how SMRM became ISMRM, and that you had quite an influential role in this. From 1982 to 1987, you served on the board of trustees, and as president from 1989 to 1990. In 1992/1993 you served on the committee overseeing the merger of the Society for Magnetic Resonance Imaging (SMRI) and the Society for Magnetic Resonance in Medicine (SMRM). What was the rationale for the merger?
JI: There were several reasons. I think the primary motivation was money. There was significant pressure from the MR manufacturers to avoid setting up for two meetings. Clearly, many people went to both meetings, (not me because I was a spectroscopist) – and so it made sense.
The merger was a difficult task because the two societies had very different models for how to do their business. SMRM did everything in house, and we had a staff who had competencies in administration, publishing, and running meetings, while SMRMI outsourced everything. Ultimately, the SMRM model was chosen for ISMRM. Moreover, the executive director of SMRM at that time, Jane Tiemann, was chosen to be executive director of ISMRM. I’m very glad that the two societies merged, and that efforts were made to not disenfranchise the pure spectroscopists. Spectroscopists are not only a minority, we are a very, very small minority.
CONTRIBUTIONS AS PRESIDENT
DKJ: We already discussed your role as president. In 1995, you were made fellow of the ISMRM, and in 2001, you received the silver Distinguished Service Award from ISMRM. Only 20 of those have ever been awarded in the history of the society.

JI: I was given that award for the for the administrative stuff, not for my science. You have to get a gold medal for the science.
DKJ: Well, that must have been a very proud moment?

JI: Yes, the silver medal was wonderful, because it recognised my administrative contributions. When I was president, we were undergoing significant growing pains. We were in the process of becoming larger, more well-known and more established, and we discovered that we had no written policies and procedures for anything. So I spent a great deal of that year flying back and forth to the Berkeley office and creating the policies and procedure manuals that served as the basis for the merged societies. Working closely with Herb Kressel, I also developed the overall administrative structure still used by ISMRM.
The other thing I did was to develop a strong executive committee. There were times for months on end where we would meet every week over phone. It’s very important to build consensus. That was also part of the transition from being a “Mom and Pop” organization to a more professional organization.
I also introduced the Young Investigator Award. That’s just a no brainer. It really helps the people who are the finalists gain profile, and that helps them with their career development.
DKJ: Well as a runner up in the Young Investigator Award. I can fully agree with you. It was a career booster.
JI: What was the title of your project?
DKJ: “Smashing Pumpkins and Squashing Peanuts”. It was about how noise distorts the profile of diffusion in fibers. Yaniv Assaf, who I’d already become friends with, was also in the final and we sat mutually disappointed, side-by-side, as the prize was given to the more-deserving Florian Wiesinger for his work on parallel imaging. The right person won.
JI: But all three of you were at least profiled and you made friends, so that’s good. The very first winner was Doug Lewandowski, who was immediately recruited to the MGH in Boston.
DKJ: That’s an awesome legacy, to have set that wheel in motion!
CARDIAC ENERGETICS
DKJ: So I’d now like to talk about the science. You’ve been a prolific writer, with over 140 papers focusing primarily on cardiac energetics. For the reader who is unfamiliar with basic physiology, could you just give us a quick summary of the kinds of questions you’ve been interested in? Perhaps starting with ATP, which, in one of your review articles, you refer to as the “universal currency of energy”.
JI: My own passion is indeed the energetics of the heart, or muscle, basically excitable tissues, but the heart is the easiest model to study. Okay, no living organism, no cell can live without the chemical energy derived from ATP. As you say, it’s the universal currency of life. That’s not my phrase, unfortunately, I wish it were. Very famous biochemists before me called it that. If you were a student of Lehninger at Hopkins, you think Lehninger invented it, and if you were a student of Stryer at Stanford, you’d think he invented it. The concept of there being a single small molecular weight molecule that is responsible for supplying the chemical energy needed for all processes in the cell is around 80 years old. So if one wants to study something important, what could be more important than the basis of life?
DISCOVERING 31P NMR SPECTROSCOPY FOR THE HEART
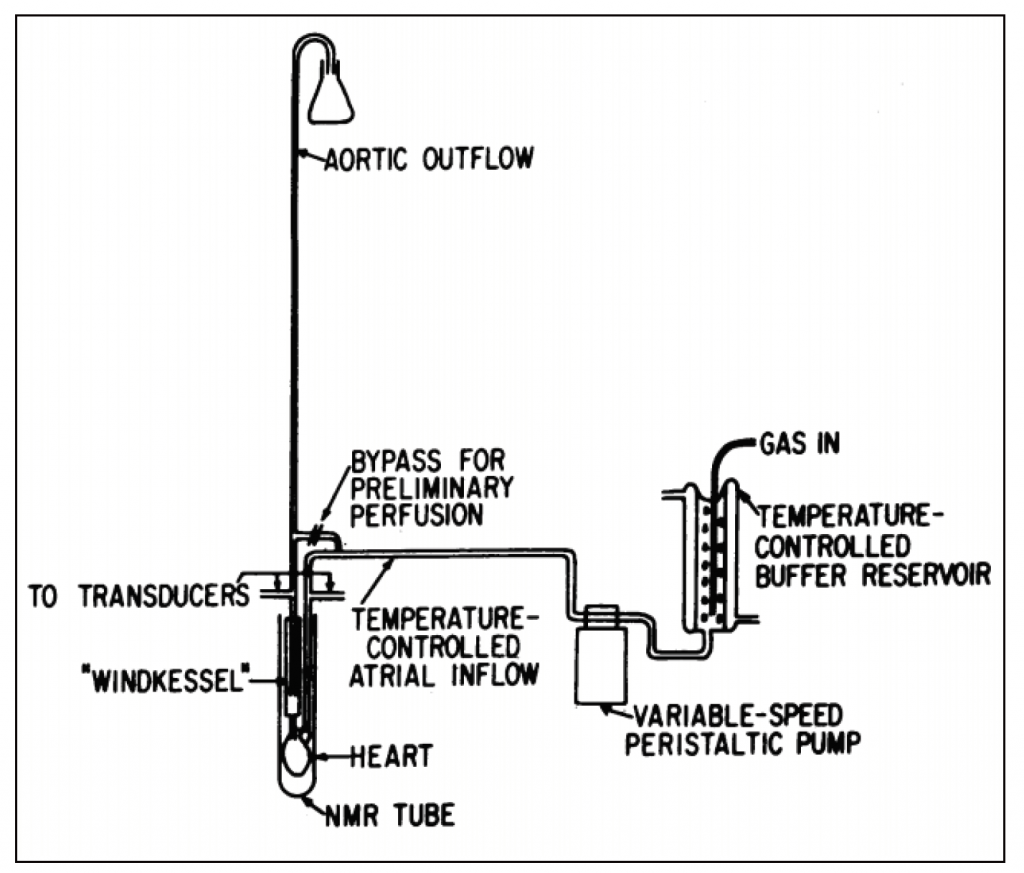
JI: There are lots of ways of studying ATP. But the only one that’s any good is NMR! Before NMR spectroscopy, to get a handle on whether the amount of ATP changed in a tissue or organ you had to prepare and process your sample. Let’s imagine an isolated heart hanging from some complicated plumbing that’s perfusing it. You’d have to smash it as rapidly as possible with tongs chilled in liquid nitrogen, making wafers as thin as possible. Then you have to give it to a really good technician who had the good hands to grind it up without letting it get warm and then finally perform demanding biochemical essays. We had lots of biochemical assays, but we didn’t have a good way to preserve the physiologic state of the sample we were interested in. And few pairs of hands can really prevent some loss of ATP. As I am fond of saying: NMR allows us to respect the integrity of the physiological state of the heart while simultaneously defining molecular events occurring within cells.
So – what’s not to like when you discover phosphorus NMR spectroscopy could be used to look at the amount of ATP that was in the heart, how it was being made and used – and all in real time without destroying the tissue?
DKJ: I’m fascinated to learn how you and phosphorus spectroscopy became acquainted?
JI: I had just received an award from the American Heart Association and was recruited by the cardiology group at UCSD. I was a very junior faculty member collaborating with the late Marlene De Luca, who was the more senior biochemist. She was part of the Lehninger biochemistry group at Hopkins and her husband, Bill McElroy, became Chancellor at UCSD, so Marlene needed a place to work. Totally by chance, she and I ended up in the same cardiology group at UCSD.
Well, Marlene comes into the lab one day, and says, “Look at this thing. David Hoult in England has literally just cut up a hunk of muscle and put it in an NMR tube, and you can see ATP and phosphocreatine”. Well, the muscle wasn’t contracting and so it was a stable system. It was a brilliant experiment in concept, but trivial to execute. And so we said, “We’ve got to do this for the heart!”.
So, we march over to the chemistry department and talk to the NMR spectroscopists that were used to only putting solutions in a narrow bore magnet. They didn’t want to have anything to do with physiologic systems. They thought the heart was disgusting. They had a narrow bore system so the only hearts that would go in would be fetal mouse hearts weighing two to three milligrams. But even those were too big to have adequate oxygen diffusion – so it was a bust.
We quickly figured out that we had to understand how to perfuse the heart. People down the hall said, “Oh, I can make that happen”. Well, by the time they ever got around to trying, my husband and I had moved to Boston, me to the Brigham. People there also said they could do that – they couldn’t. So we went to Hershey, Pennsylvania, to visit Bob Neely and Howard Morgan who were venerable cardiac physiologists. They taught us how to perfuse hearts. We came back and started to perfuse rat hearts in the 270 MHz system at MIT. And that’s how it all started. Then we got our own system, the first wide bore 360 MHz system in an academic environment, and we shared it with the world and also did our own work.
So it was fun to do something new, that nobody else had done. Unbeknownst to me, my colleague, Bill Jacobus at Hopkins was trying to do the same thing. And of course, he beat me to publication because I was always pretty bad at writing things.
COMPETITION
DKJ: That prompts me to ask about competition in the field at the time. Who were your competitors? And was it competitive or collegiate?
JI: Both! In the early days of NMR spectroscopy, there were two or three giants: the George Radda group, the Bob Schulman group, and Britton Chance with Jack Leigh’s incredible brilliance with technology.
It wasn’t competitive in the sense of who was racing to do the same experiment, because everybody had a different idea of what experiments were important, as we had very different backgrounds. Bill (Jacobus) and I were both trained as biophysical chemists, but we were interested in doing biophysical chemistry in intact tissue. It’s a different mindset and a different approach.
DKJ: Was the competition a motivator for you?
JI: No, we were too busy trying to survive. So many people were coming to the lab from all over the world because it was new, including people from Germany, France, Belgium, Italy, and Japan. Senior people too. Charlie Springer, “Mr. Sodium NMR” did a sabbatical in my lab and sent us two of his students. In fact, when I left the lab, it was taken over by one of Charlie’s former graduate students, Jim Balschi. Jim did his postdoc with us and was then recruited to a faculty position at Harvard. Jim has only recently retired himself. He is a very fine spectroscopist. No, I don’t think the competition was a motivator. I think I was already maxed out supporting my own work, so it was a busy time.
OTHER SCIENTIFIC MILESTONES
DKJ: You referred to the study of ATP and cardiac energetics as your personal passion. But there were other important outputs of the lab that you wanted to talk about….
JI: Yes, from the “NMR Lab for Physiological Chemistry”. The first important thing we did, at the Francis Bitter National Magnet Lab at MIT, was to figure out how to gate the NMR experiment to the heartbeat. That was important, because if you perfuse the rat heart, with a substrate that rapidly produces a lot of ATP from mitochondria, (i.e., you give the heart pyruvate), you don’t see any fluctuation in ATP or PCR across the beat. But if you deprived the heart of a substrate that it liked, and gave it only glucose, and varied the amount of work the heart did, then you could see enormous changes across the cardiac cycle. This is because it was really a stressed-out heart – like being in the middle of a heart attack. So, that was fun. A lot of people tried to repeat that experiment, some of them more successfully than others. It was repeated using freeze-clamping and standard biochemistry and the results agreed with us. So that was important.
When we first got the 360 MHz, we recruited Jean DeLayre to be the NMR person in the group. He immediately turned the spectrometer into an imaging system and obtained the first non-proton image. That’s his work. He did it all in a week, and it was published in Science. That was pretty cool.
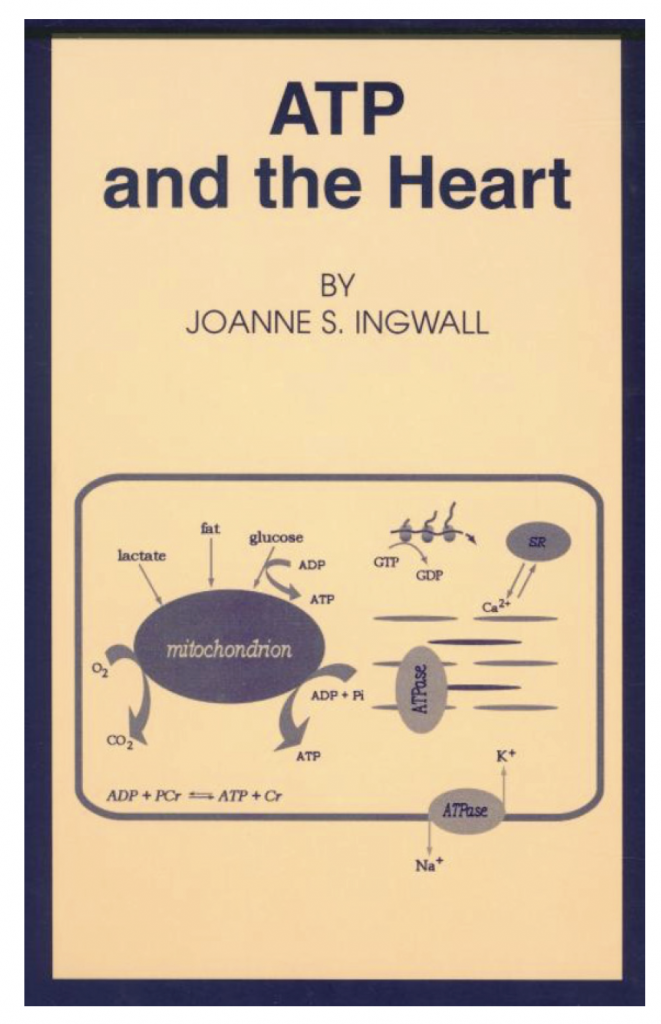
The other thing I think is really important that the lab did was working with Charlie Springer on sodium NMR of the heart. I mean, how cool is that? That you can use NMR to track the movement of an ion from outside space to the inside space in real time? “Oh, my God!” I mean, that’s so cool!
So those were the watershed projects. The fun thing for me with those experiments was that nobody else had ever done them before us. That’s the coolest part, you know? And that’s the motivator.
DKJ: So those are the things that went well. But I guess you will also learn a lot from the things that don’t go well. What were the biggest technical challenges that you had to overcome?
JI: The biggest was a simple one – and for a chemist this is really tough. We had to be sure that when we assigned a concentration, or an amount and then assume the volume, that it was in, was correct. We spent a lot of time building standard curves as you would do in any kind of chemistry experiment, that would allow us to assign a value to all the intensities of the peaks. That took a lot of time. It sounds straightforward, but it was not. We used a combination of standard curves in the NMR tube and mapping the homogeneity of the field across the NMR tube (because if we’re not homogeneous we’d be messed up). We had a really nice magnet, so that was not a problem. And then I set up a whole wet lab to do all the biochemical measurements to support the NMR experiments, not only the amounts of the metabolites, but also had machines that would measure enzyme activities, isozyme distributions of the family of isozymes. So we had to do all of that. That’s expensive and time consuming – but it’s doable and we did it.
What didn’t work? There were reports that you could use F-BAPTA and fluorine NMR to measure intracellular calcium concentrations in a heart. If only! BAPTA had to be given in such high concentration that it killed the heart. BAPTA is a chelating agent that reversibly binds calcium, so you’re using a compound that changes the size of the pool that you are trying to measure. From the ‘get go’, it was ridiculous. Two parts were bad: One it killed the physiology; and two it was bad chemistry.
DKJ: Okay, it sounds like a winner!
JI: We spent a lot of time on that. We also spent a lot of time on bioengineered mice, which turn out to be not as uniform as you would hope. You have to study a lot of mouse hearts to get any kind of believable statistics.
DKJ: It sounds like most of the challenges were around the biochemistry and perturbing normal physiology, but not so much with the NMR?
JI: Well, when Jean DeLayre was in the lab, he was such a phenomenal spectroscopist, he figured out how we could do magnetization transfer experiments. The method had been worked out by Forsén and Hoffman in Sweden in principle. In practice, when you try to apply that to a system as complicated as a heart you have to apply the saturating pulse over a relatively narrow ppm. Otherwise, you would totally mess up the experiment. We had ways of checking it because we would move the saturating pulse 300 ppm, to a region that had no signal. We built in as many checks as we could, but I’m sure the saturating pulses were not perfect. And as a physical chemist, if it’s not perfect, it’s not great. But it was the best we could do. Measuring the rate of reactions in an intact beating heart …in vivo biochemistry…is nothing but seductive. We had to do it.
ATP AND THE HEART
DKJ: You’ve highlighted the accomplishments of the lab. In addition to your papers, there’s your magnum opus, big book “ATP and the Heart”, published by Springer in 2002.
JI: My little book? That took me a year. A little bit of it was written by Jim Balschi, I have to say, and all the diagrams were all done by my admin assistant, Linda Johnson, who took care of me for 30 years, and is still my friend.
THE ROUTE INTO SCIENCE
DKJ: I’d like to go back to the early years. What inspired you to follow a career in science? Did you always want to be a scientist growing up?
JI: Yes, the only debate was whether I’d go to medical school first or go to graduate school. I guess I always had an interest in physiology. The neighborhood boys would catch frogs in the pond for me to cut up, much to my mother’s horror and my father’s delight!
I was a chemistry major in my undergraduate school, in part because the scholarship for studying chemistry was greater than one for studying biology. I like chemistry, it’s the stuff of life, and biochemistry is truly the stuff of life. For graduate studies, I went to the Chemistry Department at Cornell University and obtained a degree in biophysical chemistry in Harold Scheraga’s laboratory. Harold is an eminent structural protein chemist. But my project in the lab was not peptide structure, it was to study a blood clotting protein, a side interest of Harold’s. I was in the cold room all the time isolating prothrombin from blood. When I got a good enough preparation, I put it in all the biophysical instruments that you could put it in. That was my thesis, and it was the first physical characterization of a blood clotting protein.
So I already was interested in proteins, but I didn’t want to be in the cold room anymore. I then went to UCSF, and, spent some time on myosin, which was very messy. At that point, nobody even knew how many subunits there were. I got interested in cells and how they make proteins. Then we moved to UCSD and I started collaborating with Kern Wildenthal who was, at the time, an investigator at Southwestern Medical School (he subsequently became its President). I learned how to culture fetal mouse hearts from him and use them as a model to study biochemistry and physiology of heart diseases and cardiac development. So my interests just evolved. Some people choose what they want to study very early on and never change. I moved around.
DKJ: Your recent focus over the last ten years has been on transgenic animals. One question that often gets asked is about translatability. In your opinion, are the animal models a good model of human physiology, or have there been some surprises when one attempts to translate a finding from a rodent heart to a human heart?
JI: Well, there must be some surprises, but in my experience, we learned things from the transgenic mice well before they could be shown in the human. Maybe I was lucky in my choices. The example that I can give you is a collaboration with Kricket and John Seidman, who are well known in the field of familial hypertrophic cardiomyopathy. Kricket was the first in the world to discover that a single amino acid mutation in myosin was associated with a cardiac disease in humans.
Everybody thought that was going to explain heart failure. Well, that didn’t happen. But they made a mouse to mimic the disease. We put the hearts in the magnet, studied the energetics and physiology and found that it displayed pure diastolic dysfunction. Systolic function was normal. It took years before they could show that in humans, but they reproduced our mouse experiments.

DKJ: That leads me to ask about clinical adoption of MRS. At the 2014 ISMRM, there was a closing plenary session, because we were in Milan, it was given the title, Bamboccioni, which is a name for a bachelor who hasn’t left the home yet. The allusion was to powerful MR techniques that have yet to leave home, and the challenges to widespread clinical adoption. MR spectroscopy was included in the talk, as it seems to always be just around the corner from being widespread adopted.
My question to you is: “Are we there yet with widespread clinical adoption of spectroscopy?” And I think your facial expression says it all! If not, why not? And what will it take?
JI: You really should ask Carolyn Mountford in Australia. She was one of the women early in MR. So that’s how we met and ultimately became friends. When she was at the Brigham and MGH, she spent all her time on molecular imaging. I probably don’t know enough to answer your question, but I can tell you my gut feeling. They’re not easy experiments to do in humans. They’re really hard technically. How do you fix the localization issues? It may be that I don’t know whether the localization issues have been solved, or whether they’re being solved or whether anybody’s even trying harder to make that happen.
It’s probably not a money-maker. It may be wonderful in terms of understanding the biochemistry and coupled with the physiology of a disease state or something about disease prediction and progression, but I bet the scans take so long that it’s financially not a money maker yet, and maybe never will be.
DKJ: So are there other outstanding challenges in spectroscopy or phosphorus spectroscopy? I’m thinking about inspiring the next generation, telling them “If you could crack this, would it make a big difference”?
JI: Mitochondria have now been elevated in terms of popularity, all over the world. And I think in the next five to ten years, using spectroscopy as well as other tools, we will see some major new insights into how mitochondria work and are regulated.
DKJ: So maybe that will be the target for someone’s Young Investigator Award in the future!
PROMOTING WOMEN IN SCIENCE
DKJ: I wanted to move away from the science for a moment and explore a couple of other topics. Looking at your CV and the accolades that you’ve received, one thing is very striking. You were the first female president of SMRM, there were just two women on the original MRM editorial board, and you’re the only female recipient of the distinguished silver medal.
But I wondered if you had reflections on changing diversity and promotion of women in science. Was it important to you that you were a rare female scientist in such positions of leadership in the field at the time?
JI: No. I didn’t have enough time to waste worrying about that stuff. I had too much to do. There was a period of time, 15-20 years ago, where all the teaching hospitals at Harvard Medical School were creating offices for women’s careers. So I’m thinking this is really dumb. They should have offices for faculty careers – everybody’s careers, men and women, PhDs and MDs.
When Herb Kressel moved to the Beth Israel Deaconess Hospital at Harvard, as head of radiology, we wanted to work together. We had already worked side by side, as president and vice-president of SMRM. Herb invited me to come to work with his faculty, all MDs, all radiologists, to see if I could increase the success rate of their grant writing and their rates of promotion. The bizarre experiment of matching a PhD with MDs worked!
I learned that the Beth Israel Deaconess was going to copy the MGH and the Brigham models and create an office for women’s careers. I was asked to apply but I said that I would not take such a job unless it was for everyone. So they changed the focus, and I took the job. I think that’s a better strategy. It’s not just all boats rising, but it’s a better strategy for the institution, for the culture of the hospital. Similarly, later on, when the Department of Medicine at the Brigham and Women’s Hospital created an office for faculty careers for everyone, I led that.
MENTORING
DKJ: You have won a number of awards for your role in mentorship, including the John MacArthur Research Service Award from Brigham and Women’s in 1999, and the A. Clifford Barger Excellence in Mentoring Award from Harvard in 2000. It seems that mentoring and promoting junior faculty is just as important to you as the science?

JI: Oh yes, absolutely! As soon as I became full professor, which took forever, (it’s Harvard for heaven’s sakes, I was 50), I was delighted to be able to do a number of faculty development jobs and ended up doing something at all the teaching hospitals, except Children’s.
DKJ: So, perhaps this is an unfair question, but which was most important to you, your accomplishments in mentoring or your accomplishments in science?
JI: That’s hard to answer, because in some ways, if you have helped, say 200 young people with their careers – those investments probably have a bigger payoff. On the other hand, I don’t think I could imagine not doing science. I still do science. I do vertebrate curation for the local State Park; I am past president of the local botany society. My husband and I do several citizen science projects in Massachusetts in the summer: water quality, saving terrapins, tree phenology – but they’re all “Mickey Mouse” compared to real science!
CAREER ADVICE
DKJ: So to conclude, I’d like to go right back to where we started. You have just talked about helping 200 young faculty starting out, and I started the interview by asking you about your first time at the annual meeting. If you could speak to Joanne Ingwall, that was attending her first meeting in Montreal in May this year, with 40 or 50 years of research ahead of her, what advice would you give to her?
JI: (1) Don’t be afraid to try new things; (2) Don’t be afraid to ask someone to collaborate. (3) Maybe I’m colored too much by what it took to get promoted at Harvard – but it’s probably true in most institutions – you’re not going to be promoted if you’re just doing something well that other people are also doing. You have to create your own unique contribution. And (4), get a mentor to help you navigate the political world.
And you can’t hide under a bushel. You have to invest in your own career development. I hate to say it this way, but you have to sell your science. You cannot be just somebody that sits in a little lab and doesn’t communicate. It isn’t enough to write papers, you’ve got to go to meetings, you really do! It’s a rare person who can really be a player in the field, who hates going to meetings.
DKJ: Excellent advice! Well Professor Ingwall, it has been a real honour and privilege spending time talking with you today. On behalf of the entire Highlights team, I’d like to thank you for your scientific contributions, for your mentoring contributions and for your contributions to the Society. But most of all, thank you for taking the time to talk to Highlights!
JI: Well – thank you. It was a pleasure!